Genetically Modified Organisms
Transformation of plants and functional genomics

Expression of proteins visualized by fluorescent markers. Photo: David Dobnik
Sedanje raziskovalno delo je usmerjeno na analiziranje izražanja genov v različnih linijah transgenih rastlin krompirja, ki se pojavijo kot odgovor na okužbo z virusom PVYNTN. Analizo izvajamo s pomočjo tehnike mikrorastrov in analize z metodo PCR v realnem času. Primerjamo izražanje genov med odporno in občutljivo transgeno rastlino.
Development of methods and approaches for detection of genetically modified organisms (GMO)
Each year new genetically modified organisms (GMOs) are entering the world market challenging existing testing schemes. Therefore, new approaches and methodologies have to be developed. Classical scheme using molecular methods targeting genetic elements specific to GMOs is shown below. Main phases being: screening for genetic targets common in as many GMOs as possible, identification of individual GMOs and further quantification of approved GMOs.
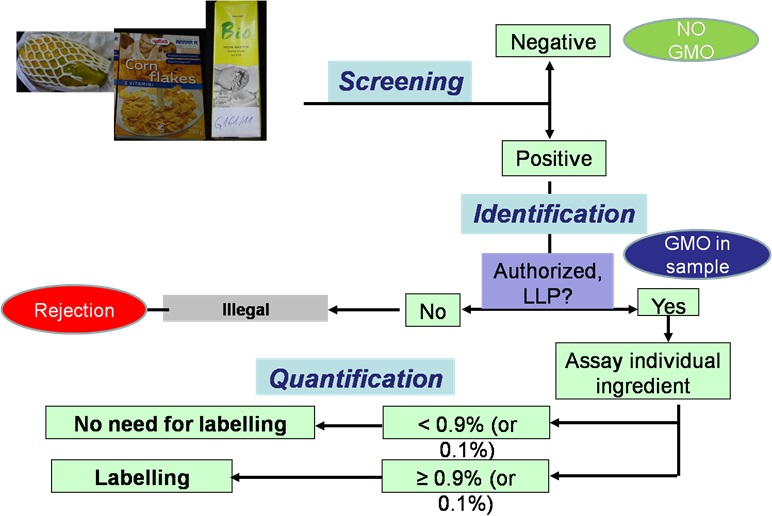
Scheme for GMO testing.
Screening phase is important to cover as much as possible GMOs, therefore during GMOseek and GMOval projects, headed by NIB, new methods for GMO screening were developed. Additionally, matrix based approach was developed and matrix database established. Software GMOseek was developed in cooperation with the Jožef Štefan Institute, to provide optimal or near-optimal solutions of the GMO testing process and to support the inspection of testing results. Matrix and software are freely available on the internet page of GMOseek project.
Development and application of digital PCR (dPCR), enabling absolute quantification of DNA copies, for identification and quantification of GMOs as well as for validation of methods, is one of the hottest topics. We were the first using digital PCR (Bio-Rad QX100™ ddPCR™ System) for GMO analyses (MORISSET, Dany, ŠTEBIH, Dejan, MILAVEC, Mojca, GRUDEN, Kristina, ŽEL, Jana. Quantitative analysis of food and feed samples with droplet digital PCR. PloS one 2013, 8, 5, e62583-1-e62583-9. doi: 0.1371/journal.pone.0062583).
Further development of multiplex dPCR is in a process especially in the frame of DECATHLON project (7FP). This project brings together a broad range of experts and expertise to jointly work on the development of new or improved methods that are needed in the field of 1) food pathogens, 2) traceability of GMOs and 3) customs issues. NIB is leading work on digital PCR for GMO detection and development of isothermal methods, mostly for detection of Escherichia coli.
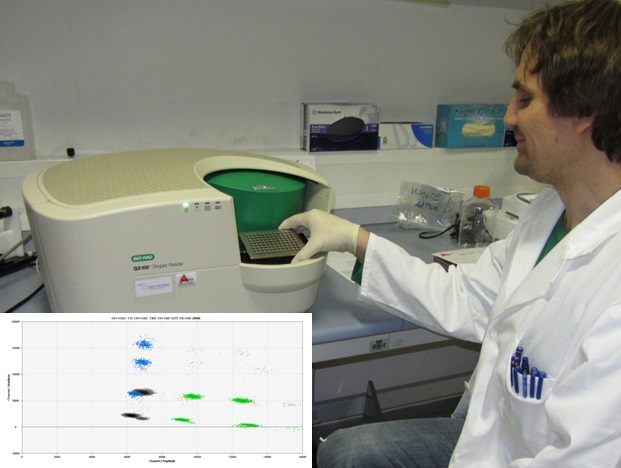
Digital PCR is becoming important part for molecular analyses in different fields of research and applications.
New high throughput and cost efficient methods are main challenge for recent GMO detection. They will be further developed in bilateral project with Chinese partner Schanghai Jiao Tong University.
National reference laboratory for GMO detection
As National reference laboratory for GMO detection, nominated by responsible Ministries, we cooperate intensively with European Union Reference laboratory for GM food and feed. We are involved in validation of methods for identification and quantification of new GMOs entering the European Union market. NIB is a member of the European Network of GMO Laboratories (ENGL), where we have representative in Steering committee and we actively co-operate in expert working groups. In our laboratory studies of characterization of reference materials are done for the Institute for Reference Materials and Measurements (EC JRC IRMM). By IRMM we are validated also as a supplier for characterisation studies with digital PCR.
At the national level we offer professional support to all ministries and inspection services, which are competent for GMOs (Ministry of environment and spatial planning, Ministry of agriculture, forestry and food and Administration of the Republic of Slovenia for Food Safety, Veterinary Sector and Plant Protection) as well as companies needing scientific advice related to GMOs. We are testing GMOs in food, feed, seeds and plants for official control.
National biosafety System
During establishment of biosafety system in Slovenia we were active partner to the Ministry of environment and spatial planning. Current activities are focused especially on detection of GMOs, where Ministry of environment and spatial planning nominated NIB to the Electronic Network of Laboratories and Online Discussion Groups on the Identification of Living Modified Organisms. The document Technical Tools and Guidance for the Detection and Identification of LMOs was developed and is ongoing activity. NIB is participating especially on the Minimum performance criteria for sample handling, extraction, detection and identification methodology.
Our co-workers are members of two scientific committees for the risk assessment of GMOs (Scientific committee for work with GMOs in contained use and the Scientific committee for the deliberate release of genetically modified organisms into the environment and putting products on the market).
Accreditation according to ISO/IEC 17025
Metrology - bioanalyses
National Institute for Biology, Department of Biotechnology and Systems Biology is nominated by the Metrology institute of the Republic of Slovenia as a holder of national etalon for amount of substance / food of plant origin. The nomination was granted to the excellent work on analyses of nucleic acids, especially for detection of GMOs and microorganisms and ensuring traceability to lower metrological levels.
We are active member of Working Group on Bioanalysis, of Consultative Committee for Amount of Substance – Metrology in Chemistry and Biology (BAWG CCQM) and we cooperate key comparison and pilot studies in the field.
Our metrological research activities are mostly based on our wide experiences on detection and quantification of nucleic acids and validations and verification of methods. Methodologies used in this the projects are qPCR, dPCR and NGS.
We are currently participating in two projects in the frame of European Metrology Research Programme (EMRP). Joint research project "Metrology for monitoring infectious diseases, antimicrobial resistance, and harmful micro-organisms" (INFECT-met), aims to develop novel measurement procedures and validation frameworks to support current and emerging molecular approaches for efficient, harmonised and rapid diagnosis, surveillance and monitoring of infectious diseases, with a particular focus on respiratory. The ultimate aim is to establish routes for improving the accuracy, robustness, comparability and traceability of measurements within the metrology and diagnostics/epidemiological communities across Europe.
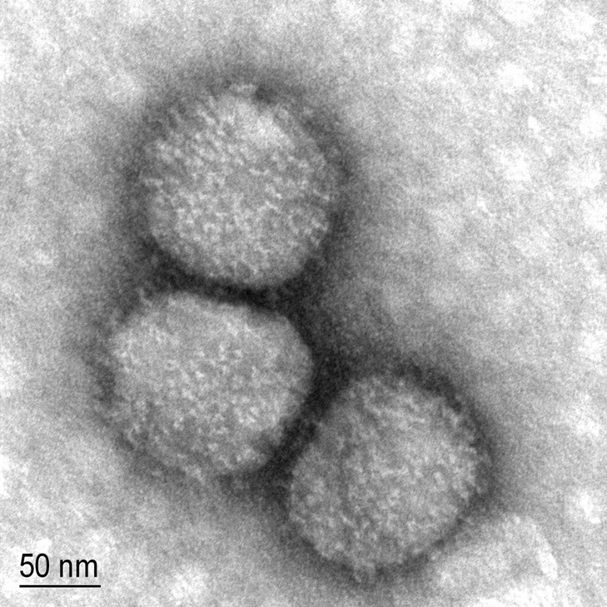
Human cytomegalovirus (CMV), a member of the family Herpesviridae, is causative for a wide range of diseases. Foto: dr. Magda Tušek.
Joint research project "Traceability for biologically relevant molecules and entities", (BioSITrace), dealing especially with accurate counting of biological entities.